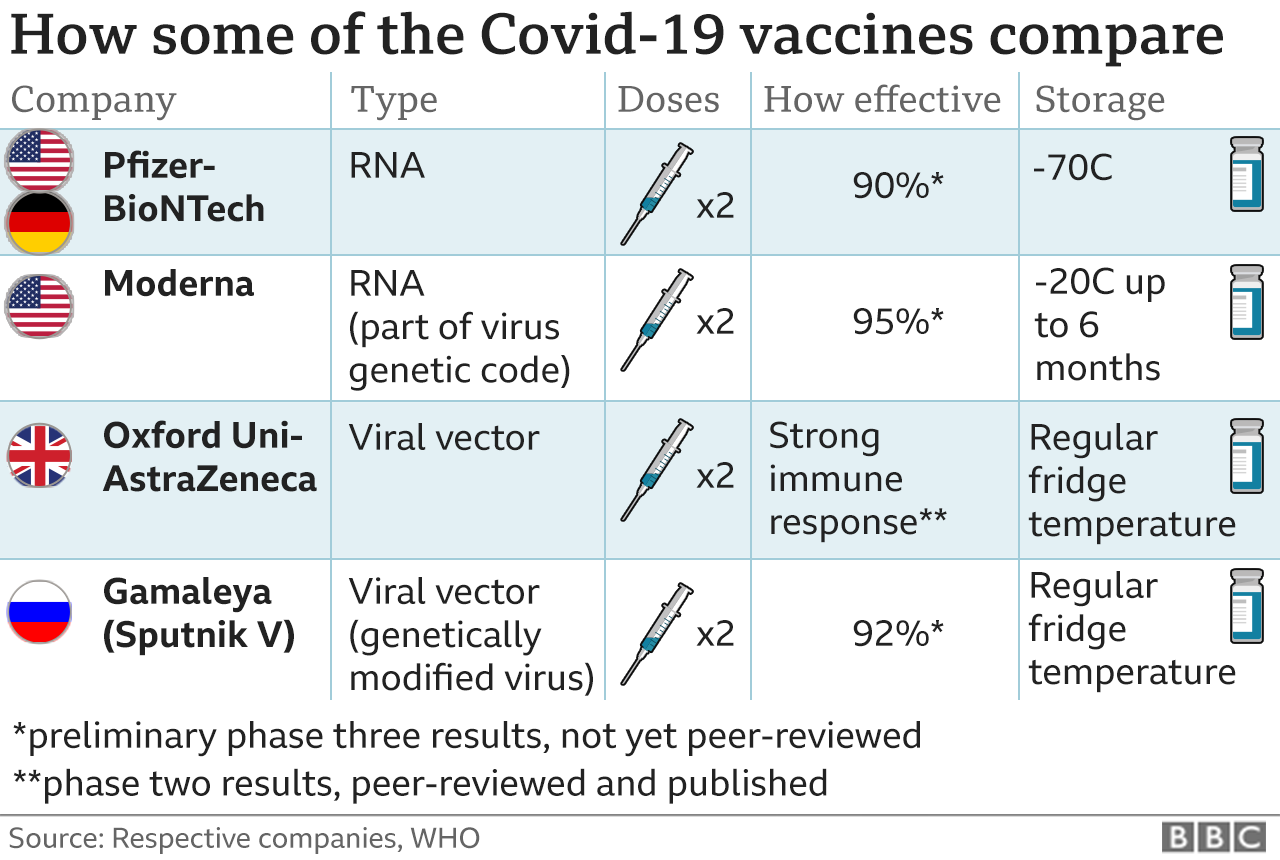
federal law, that “once a license is obtained, the data and information of a biological product in the process of license approval can be immediately disclosed to the public,” which is in line with the FDA’s commitment to “complete transparency” before and after the approval of the new crown vaccine The opposite. The plaintiffs argued that the FDA violated U.S. The PHMPT group was furious: by 2076, many of the people who made, approved, and vaccinated the vaccine had long since left the world. In November last year, the PHMPT organization asked the FDA to fully disclose the vaccine review data, but the FDA did not respond prompting the lawsuit against them.įollowing the FDA’s defeat, the court mandated them to release a 329,000-page review document for Pfizer’s new crown vaccine by this summer the FDA has now released the first batch of the documents.Ī total of 150 documents are currently published on the FDA’s website for free download but they have not released a statement to address the latest development.Īccording to our source The FDA petitioned a federal judge to disclose only 500 pages of important documents a month. hence, it will take 55 years to fully disclose the 329,000 pages of information, until 2076. District Court for the Northern District of Texas last September, Questioning the FDA’s decision to withhold data related to Pfizer and BioNTech’s 2019 COVID-19 vaccine . 
Food and Drug Administration (FDA) lost the lawsuit filed against them by the Public Health and Medical Professionals for Transparency (PHMPT), a nonprofit organisation who first filed a lawsuit against them in the U.S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
